Bacterial, viral and fungal diseases on Phalaenopsis and other orchids
Even in modern production plants and when strict hygiene measures are observed, infection of the plants with viruses, bacteria and especially fungal diseases cannot be completely ruled out. Especially infections of Fusarium oxysporum may lead to existential losses in the production of potted products and young plants. The same applies to bacterial infections with Erwinia and Acidovorax (Pseudomonas). In the following, the economically most important pathogens are listed, including their biology, damage pattern and control options. Recommended chemical control methods must be checked against the applicable approval regulations of your country. All plant protection recommendations are made with the exclusion of any liability.
Bacterioses
Biology
Erwinia caroto¬vora subsp. carotovora [Jones] Bergey et al
Erwinia cypripedii [Hori] Bergey et al
Acidovorax avenae subsp. cattleya [Pav.] Will. (=Pseudomonas cattleya [Pav.] Savul.)
Bacteria are unicellular rod-like organisms that move actively in the water with the help of flagella. Reproduction is done by division and only takes a few minutes in a humid and warm environment.
Bacteria are not able to infest a healthy plant surface, they always need open parts such as wounds or gaps.
Most bacteria reach their optimum in high temperatures between 25°C and 30°C and high humidity, so repeated explosion-like infestation usually has to be expected during the summer. But even in the darker and cooler seasons they can cause severe damage to orchids, even if the pest develops considerably slower.
Erwinia cypripedii [Hori] Bergey et al
Acidovorax avenae subsp. cattleya [Pav.] Will. (=Pseudomonas cattleya [Pav.] Savul.)
Bacteria are unicellular rod-like organisms that move actively in the water with the help of flagella. Reproduction is done by division and only takes a few minutes in a humid and warm environment.
Bacteria are not able to infest a healthy plant surface, they always need open parts such as wounds or gaps.
Most bacteria reach their optimum in high temperatures between 25°C and 30°C and high humidity, so repeated explosion-like infestation usually has to be expected during the summer. But even in the darker and cooler seasons they can cause severe damage to orchids, even if the pest develops considerably slower.
Damage
Erwinia carotovora and Erwinia cypripedii are soft rot pests, they cause soft hazy yellow spots on the leaves.
Decomposition usually starts at the base of the leaf; in humid conditions it spreads across the entire leaf leaving nothing but a pulpy mass. On the infested areas a yellow bacterial slime often occurs. When the disease
reaches the apical point, it spreads to other leaves and finally the whole plant rots.
Bacterioses on young plants in high temperatures, bad light conditions and high humidity can occur during the
whole year, but mainly during the winter months and can make the plant die within several days.
Erwinia sp. has a very broad spectrum of host plants, among which nearly all orchid varieties are found.
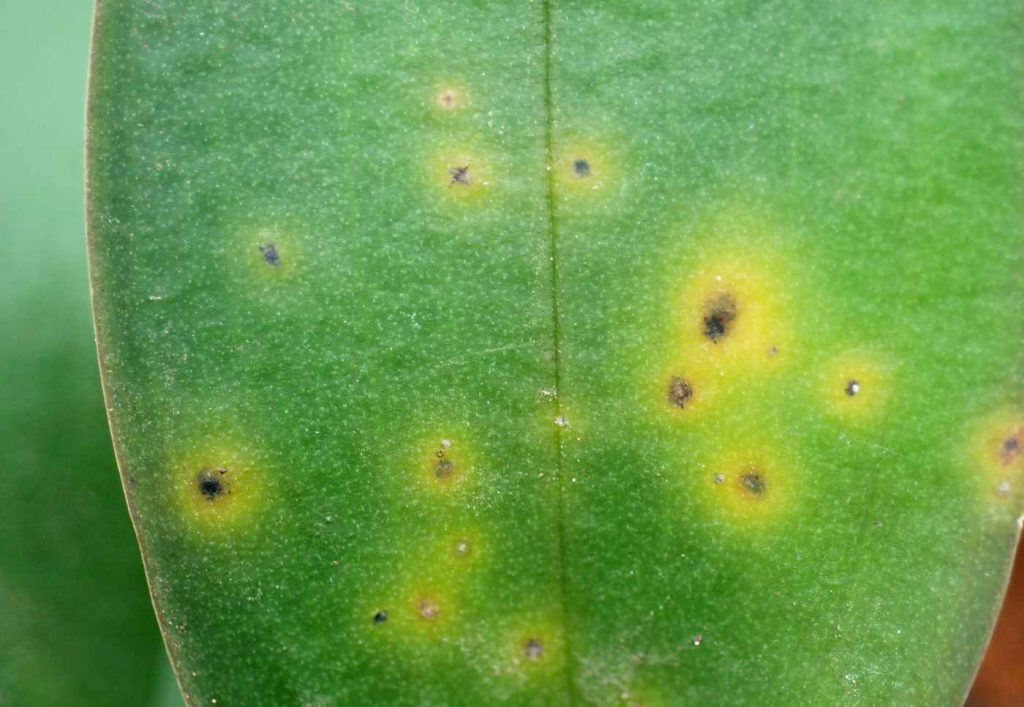
Acidovorax mainly occurs on Phalaenopsis and Cattleya. However, Cymbidium, Dendrobium, Zygopetalum and
Epidendrum are also mentioned as being host plants. At the beginning of an infestation with Acidovorax usually small dark spots appear on the leaf blade. With a backlight a marked yellow circle around the infection can
be seen. Small spots unite to form larger spots and the tissue shrinks while turning black. Decomposition can
be limited to individual leaves or spread across the entire plant. In contrast to infections with the anthracnose
pathogen Colletotrichum, no ring-formed grouped fruit bodies are found in the infested areas. Infestation with
Acidovorax may also occur on blossoms.
There may be several bacterioses on orchids at the same time. Usually the degree of infestation of the host plant is therefore not directly connected with the respective bacterium, but rather with the conditions of cultivating and climatic conditions and the condition of the plant. Thus, only microbiologic examinations can reveal the type of bacterium that causes a certain disease.
There may be several bacterioses on orchids at the same time. Usually the degree of infestation of the host plant is therefore not directly connected with the respective bacterium, but rather with the conditions of cultivating and climatic conditions and the condition of the plant. Thus, only microbiologic examinations can reveal the type of bacterium that causes a certain disease.
Control
Bacteria cannot be controlled directly. Severe adequate cultivation and hygiene measures must be taken, especially against Erwinia during the darker winter months and against Acidovorax in high temperatures in the summer to prevent infections spreading.
- Water is the most common source of spreading bacterioses on plant populations. The plants must be watered carefully, keeping the leaves as dry as possible, the plants should not be placed too close to each other.
- Wounds are open doors for bacterioses and must be avoided.
- All plants with visible symptoms must be removed from the greenhouse immediately and destroyed. Cutting off individual infected leaves hardly improves the situation and only slows down the course of the disease. In the case of an infection in young thinned out plants, the whole box should be removed.
- Stress situations such as marked temperature or light changes or increased nitrogen fertilisation have to be avoided at all costs.
- Cultures should be cultivated as regularly as possible.
- Hygiene must have top priority for all cultivation: washing your hands, changing or disinfecting work materials.
- Cultivation vessels, shelves, greenhouse desks and tools that were in contact with infected plants or water must be disinfected.
- In order to decrease spreading of the intruder in the stand, regular and repeated prophylactic spraying with copper-containing products can be done. Copper hydroxide or copperoxichloride crystals have the effect of mechanical barriers on the plant surface. However, treatment with copper over a longer period might result in phytotoxity on leaves and air roots.
- Common benzoic acid can also be used for disinfection in gardening when carefully poured over the plants, however, this product may not reach the roots. Most other disinfectants are phytotoxic and are not suitable.
- Excellent results are achieved using chloride dioxide. As an oxidating agent, it takes effect on the organic parts of the water. Chloride dioxide can be mixed with the water for watering.
Botrytis
Biology
Grey mould, blossom rot, Botrytis rot
The causative organism of leaf and stem mould and marked blossom rot on Phalaenopsis and Cattleya is the weed mould Botrytis cinerea Pers. This common intruder has a very broad spectrum of host plants; in fact it grows nearly on all parts of the plant. A characteristic feature is the grey sporal area on the infested spots (grey mould) which can be seen in high humidity. When slightly touched or due to air movement, the spores detach from their carriers and spread in the greenhouse. In favourable conditions Botrytis can also form dark sclerotical diseases.
The causative organism of leaf and stem mould and marked blossom rot on Phalaenopsis and Cattleya is the weed mould Botrytis cinerea Pers. This common intruder has a very broad spectrum of host plants; in fact it grows nearly on all parts of the plant. A characteristic feature is the grey sporal area on the infested spots (grey mould) which can be seen in high humidity. When slightly touched or due to air movement, the spores detach from their carriers and spread in the greenhouse. In favourable conditions Botrytis can also form dark sclerotical diseases.
Damage
At first the blossoms show small watery spots that can be seen with a back light. Later they
become brown spots and the tissue becomes necrotic. Symptoms mainly occur in constantly
high humidity and water sprinkling, resulting from conditions below dew point. Dirty greyish
spots may occur on the leaves and on these spots, the dusty sporogenesis will appear.
Control
- Botrytis is a secondary parasite; well nourished and optimally cultivated plants will hardly be infested.
- Insufficient light, unbalanced nitrogen fertilisation or too high a salt content in the substrate increase the susceptibility of the plants.
- During blossom formation it has to be ensured that the conditions are not below dew point to avoid the resulting water sprinkling from damaging the plant. Air humidity must be decreased especially in winter, stagnating air has to be avoided. In some cases the use of fans may be recommended.
- In non blossoming stands repeated prophylactic treatment may be done with fungicides. As Botrytis becomes resistant to fungicides very quickly, the agent groups have to be changed constantly.
- Direct chemical control of Botrytis blossom rot does not make any sense.
Colletotrichum
Biology
Anthracnose
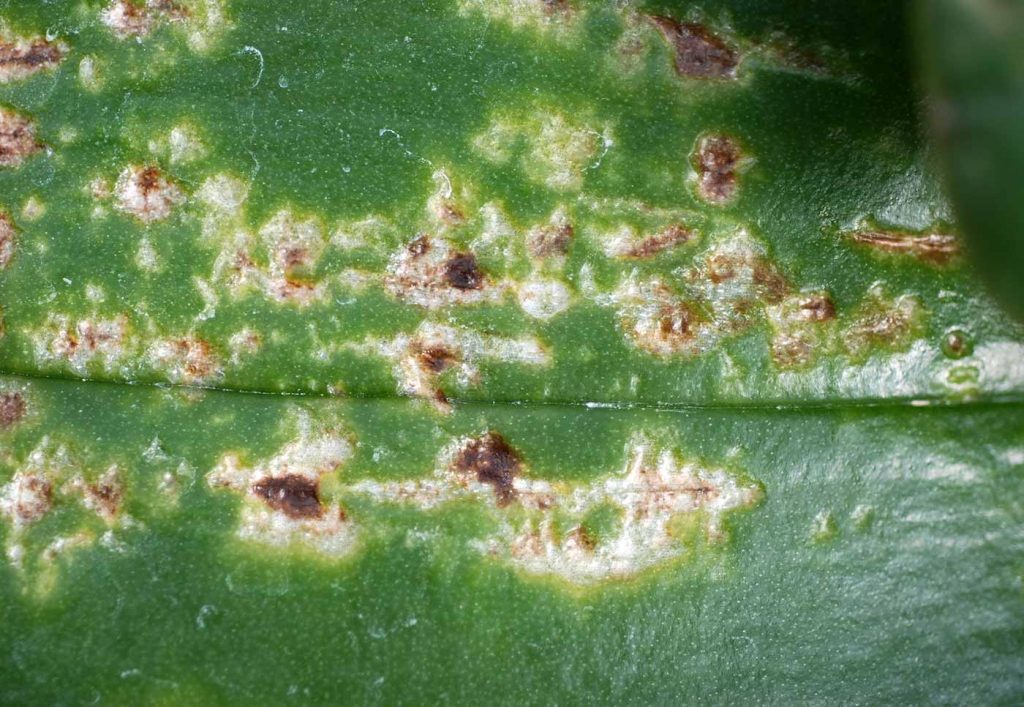
Anthracnose on orchids is caused by the weed mould Colletotrichum gloeosporioides Penzig (= Glomerella cingulata [Ston.] Spauld et Schrenk). It appears in glasshouses when humidity is high or when the leaves have been wet for a long time. Circular spore deposits are found on leaves and the spores are spread in the stand with constant high humidity or wet leaves. Water sprinkling is the most common form of spreading. Infection via the roots is not possible.
Anthracnose on orchids is caused by the weed mould Colletotrichum gloeosporioides Penzig (= Glomerella cingulata [Ston.] Spauld et Schrenk). It appears in glasshouses when humidity is high or when the leaves have been wet for a long time. Circular spore deposits are found on leaves and the spores are spread in the stand with constant high humidity or wet leaves. Water sprinkling is the most common form of spreading. Infection via the roots is not possible.
Damage
Small round, brown spots mainly occur on the leaves of Paphiopedilum, Phalaenopsis,
Zygopetalum and Masdevallia. These spots can be clearly distinguished from the healthy
tissue and may form shiny black spots when coalescing. With the disease proceeding, spore
deposits form on the destroyed tissue in concentric circles, excreting slimy pink to yellowish
spore masses. If the disease reaches the stem base, the plant may die prematurely.
Anthracnosis spots appear on the pseudobulbs of Odontoglossum.
Control
- If there is a risk of infection, air humidity should be decreased. Watering from above should always be avoided, at least drying off the leaves should be carried out immediately.
- When there is only slight infestation at the beginning, infected leaves can be cut off, markedly infested plants have to be destroyed.
- Prophylactic, repeated treatment with organic fungicides is possible.
- When symptoms are detected, spraying with fungicides that are effective against leave spotting diseases should be carried out.
Fusarium
Biology
Fusarium rot - stem and root rot
The main pest on Phalaenopsis and Paphiopedilum is Fusarium oxysporum Schlecht. The fungus forms its fungal spores on the usually whitish to pinkish spore hymenia (sporodochiae). They are either long and sickle-shaped and serve to survive (macro conidia), or they are small, globose to oval (micro conidia). The conidia spreads the infection in the stand and infests further host plants. Most parasitic Fusaria are vascular parasites, they obstruct the conducting tissue and cause wilt diseases. They are typical secondary parasites, the reasons for infestation can be too high a salt concentration, low ground temperatures, gnawing by organisms in the ground and inappropriate substrates that are too wet or contain too much peat. Fusaria and other fungi multiply particularly quickly in sterile plant substrates with a high percentage of peat, because in those materials there are no other micro-organisms as natural antagonists.
The main pest on Phalaenopsis and Paphiopedilum is Fusarium oxysporum Schlecht. The fungus forms its fungal spores on the usually whitish to pinkish spore hymenia (sporodochiae). They are either long and sickle-shaped and serve to survive (macro conidia), or they are small, globose to oval (micro conidia). The conidia spreads the infection in the stand and infests further host plants. Most parasitic Fusaria are vascular parasites, they obstruct the conducting tissue and cause wilt diseases. They are typical secondary parasites, the reasons for infestation can be too high a salt concentration, low ground temperatures, gnawing by organisms in the ground and inappropriate substrates that are too wet or contain too much peat. Fusaria and other fungi multiply particularly quickly in sterile plant substrates with a high percentage of peat, because in those materials there are no other micro-organisms as natural antagonists.
Damage
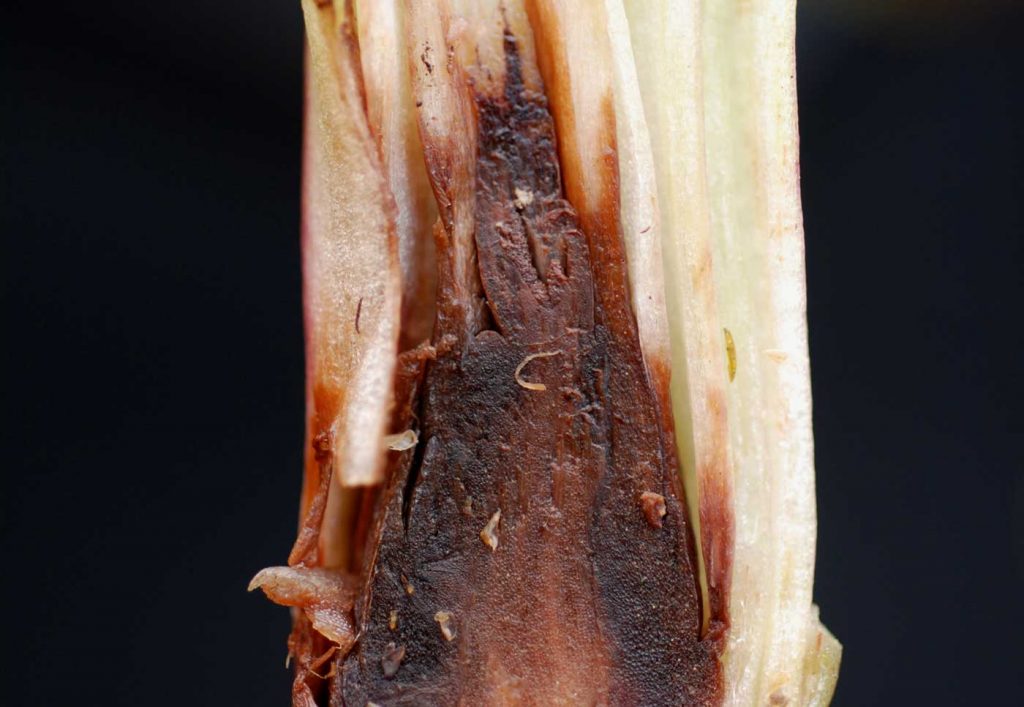
Infections caused by Fusarium on Phalaenopsis and similar hybrids are characterised by
small yellow-brown to reddish lesions on the roots. The coloured areas increase in size as
patches of dry rot appear with its typical constrictions. At the neck of the root the tissue turns
hard and black with dry rot. With increasing infestation the dry rot areas at the base of the
leaves become larger and in high humidity pink hymenia form on the dry rot areas.
The youngest leaves of Phalaenopsis become extremely reddish, there is chlorosis formation
and finally the leaves fall off and the plant begins to die. Older leaves often become
coriaceous and dry out. In some varieties, Fusarium can also damage the flower stalk
producing sunk-in, dry rotten spots, the blossoms fall off prematurely.
In Paphiopedilum, infection often affects the root insertion places and the leaf basis. Watery
rot appears, the individual leaves can then often be pulled out of the leaf sheath.
The disease progresses relatively slow. A stand can be infested for several months without
any plants dying. The healthy part of the plant often forms a lot of new roots. But usually
marked growth depressions are a sure sign of a Fusarium infection.
Control
Fusarioses on roots are often detected too late, because in the early stages, the plants do not
show that their roots are in a bad state. They are typical secondary parasites. Plants with
retarded growth due to too extensive fertilisation, exceedingly wet substrates or extremely low
ground temperatures can easily be infested. Gnawing of the roots by ground organisms also
offers favourable infestation conditions for that fungus.
- Control should be generally prophylactic by improving the respective cultivation methods.
- Severely infested plants should be destroyed or at least planted elsewhere and all dead roots have to be removed carefully.
- Treatments with fungicides against Fusarium oxysporum on roots are only sufficiently effective in the early stage of the disease. Watering with fungicides are not effective enough and can negatively influence the growth of the plants. In the case of Fusarium infestation at the base of the leaves, spraying can be useful to stop it spreading in the stand and get the infestation under control.
Pythium & Phytophtora
Biology
Pythium and Phytophthora rot
The causative organs of black rot on roots, rhizomes, stems and leaves of orchids belong to the species of the Oomycetes group. Main species found on Orchids are Phytophthora cactorum Schroet., Pythium ultimum Trow., Pythium debaryanum Hesse and Pythium splendens Braun. Characteristic features of these fungi are round to lemon-shaped sporangia. This is where the flagellate zoospores are formed that makes the causative organ spread. Sufficient humidity is the decisive factor for multiplication and spreading of the fungi in the plant stand. Constant substrate humidity helps to grow fruiting structures and zoospores. Spraying or irrigation spreads the rot from plant to plant. Most Phytophthora species prefer high temperatures and sufficient humidity, many other fungi, however, grow best in cool and humid conditions. The fungi intrude their host plants with the help of specific enzymes and excrete toxic substances in them. Within a very short time infested plant parts may decompose and there will be extreme soft rot. Whereas Pythium only infests underground plant parts as a parasite, Phytophthora may occur on all plant parts.
The causative organs of black rot on roots, rhizomes, stems and leaves of orchids belong to the species of the Oomycetes group. Main species found on Orchids are Phytophthora cactorum Schroet., Pythium ultimum Trow., Pythium debaryanum Hesse and Pythium splendens Braun. Characteristic features of these fungi are round to lemon-shaped sporangia. This is where the flagellate zoospores are formed that makes the causative organ spread. Sufficient humidity is the decisive factor for multiplication and spreading of the fungi in the plant stand. Constant substrate humidity helps to grow fruiting structures and zoospores. Spraying or irrigation spreads the rot from plant to plant. Most Phytophthora species prefer high temperatures and sufficient humidity, many other fungi, however, grow best in cool and humid conditions. The fungi intrude their host plants with the help of specific enzymes and excrete toxic substances in them. Within a very short time infested plant parts may decompose and there will be extreme soft rot. Whereas Pythium only infests underground plant parts as a parasite, Phytophthora may occur on all plant parts.
Damage
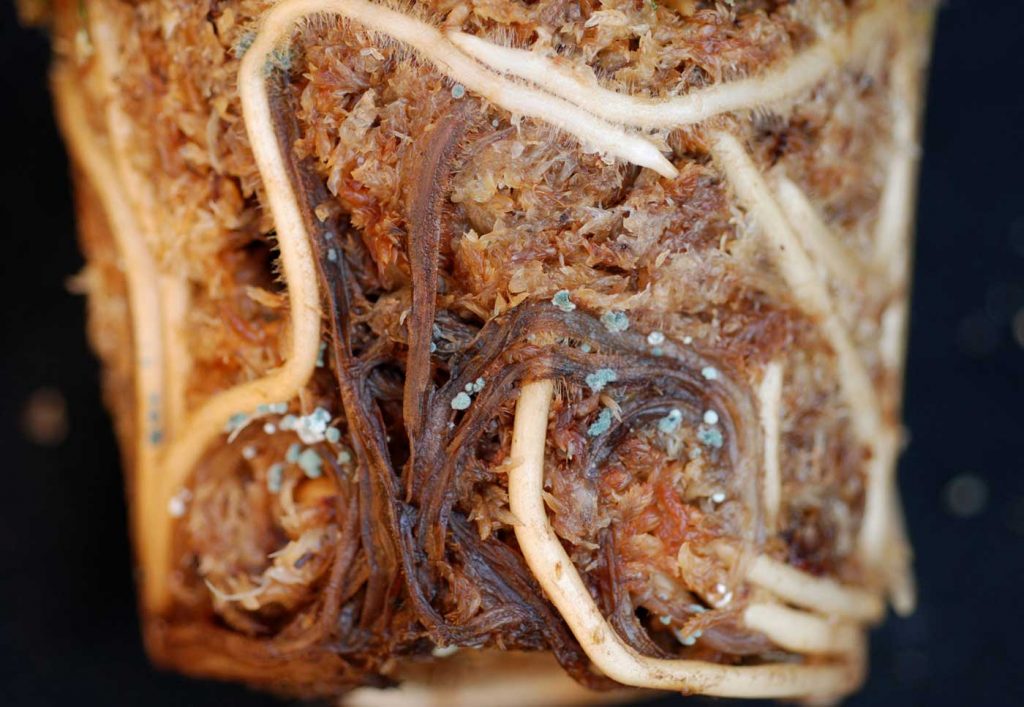
Pythium infestation on Phalaenopsis usually occurs on seedlings and young plants, less
frequently on older ones. Newly thinned out seedlings die quickly within a short time, the
roots turn brown and rot. Often the root cortex is destroyed whereas the stele is still more or
less intact. Different animal organisms feed on the roots as a consequence of or as initial
cause of the disease.
Older plants of Cattleya, Paphiopedilum, Oncidium or Odontoglossum show brown to black
infested areas on the leaf base, the pseudobulbs and the rhizomes. The infested plant parts rot
away and finally the plants die.
The disease usually occurs in the darker seasons as a result of inadequate cultivation.
Phytophthora almost exclusively infests older plants. These causative organisms that occur
frequently on orchids mainly attack the stem base, but can also infest the leaves. Quickly
enlarging brown or black rot spots occur on the infested parts which finally make the plant die
when there is extensive soft rot. Phytophthora is a causative organism that has ideal infection
conditions in high humidity and high temperatures, i.e. in the summer.
Control
- Plants showing symptoms must be removed from the stand immediately.
- Endangered stands should not be sprayed with water as this might help the zoospores to spread quickly, in companies with recycled water the pest might spread via the roots.
- Cultivation vessels, shelves, greenhouse tables and tools that were in contact with infected plants or water must be disinfected.
- To prevent infection, cultivation conditions must be improved. In addition to avoiding high temperatures (early shading required) water treatment must be done carefully. Humidity may only be increased moderately.
- Stagnant moisture is not to be expected in orchid substrates, but the substrate used for Phytophthora prophylaxis needs to meet certain requirements. The substrates used should not warm up too much, which would lead to the considerable weakening of plants in extreme conditions and contribute to spreading the disease more quickly.
- Pythium and Phytophthora belong to a special group, so direct treatment must be done with special fungicides.
Rhizoctonia
Biology
Rhizoctonia root and crown rot
The causative organism of root and crown rot on Phalaenopsis, Miltonia, Oncidium and other orchid varieties is the fungus Rhizoctonia solani Kühn. Rhizoctonia does not form any spores, but only dense mycelium settling on organic material. When the plants are more infested, there will be curling, usually at the roots. The fungus is widely spread and often gets into orchid cultivations in substrates. Wet, cold and badly ventilated substrates help the organisms to spread. In substrate, Rhizoctonia may form persistent sclerotial diseases which may be infectious for a very long time.
The causative organism of root and crown rot on Phalaenopsis, Miltonia, Oncidium and other orchid varieties is the fungus Rhizoctonia solani Kühn. Rhizoctonia does not form any spores, but only dense mycelium settling on organic material. When the plants are more infested, there will be curling, usually at the roots. The fungus is widely spread and often gets into orchid cultivations in substrates. Wet, cold and badly ventilated substrates help the organisms to spread. In substrate, Rhizoctonia may form persistent sclerotial diseases which may be infectious for a very long time.
Damage
On roots, stem bases or at the base of leaves rot areas appear showing brown fungus threads.
In high humidity, which is usually found in young plant stands, the Rhizoctonia fungus
threads grow very quickly up the plants, they are clearly seen then by the naked eye. The
leaves will subsequently show yellow-brown colour changes. The plants react by growth
stops, the youngest leaves are often deformed, they rot at the leaf base and finally drop off. As
a consequence of Rhizoctonia infestation in the young plant phase, necrotic spots may be
visible on the leaves later on.
Older plants hardly show signs of dying off, however, growth may be disturbed. The sclerotial
disease may negatively influence the structure of the substrate and the water supply due to
lump formations.
Control
- Different species of Rhizoctonia solani are found on orchids. Not all species cause damage, some are also Mycorrhiza fungi.
- Those species react very differently to the fungicides used. A change of the active principle group is therefore recommended to control Rhizoctonia in orchids. Using products to strengthen the plant such as Trichoderma harzianum or Bacillus subtilis is a good solution for young plants. These organisms are antagonists, they form deposits at the roots of the orchids and make them resistant to harmful fungi.
- When infested, the young plants should be kept drier and sprayed not more than is absolutely necessary.
Sclerotium
Biology
Southern Blight or Sclerotium rot
The causative organism of Southern Blight, Sclerotium rolfsii Sacc. (= Athelia rolfsii), is a fungus that lives on the ground and does not form spores, similar to Rhizoctonia. High temperatures help it to grow quickly, optimal temperatures are between 27°C and 30°C. The fungus needs a lot of oxygen, infestation starts in the upper substrate layers at the root neck or stem base. Roots in the substrate are not infested. Sclerotium can hardly grow in cool, humid substrates.
The causative organism of Southern Blight, Sclerotium rolfsii Sacc. (= Athelia rolfsii), is a fungus that lives on the ground and does not form spores, similar to Rhizoctonia. High temperatures help it to grow quickly, optimal temperatures are between 27°C and 30°C. The fungus needs a lot of oxygen, infestation starts in the upper substrate layers at the root neck or stem base. Roots in the substrate are not infested. Sclerotium can hardly grow in cool, humid substrates.
Damage
The upper roots and the stem base of infested plants start to rot, the rotted areas are mushy
and bad, the plants become lurid and dry. A characteristic of the disease is the quick
breakdown of the plants. In high humidity, a white to yellowish mycelium shows in the
infested areas spreading quickly in a flabelliform way. Later on dark brown round sclerotia
form in the mycelium. Sclerotia which are only about 1.0 mm in size are a major
characteristic of the disease.
Control
- Fungicide treatment is only possible in the initial phase of the disease. As soon as mycelia and small round sclerotia occur, direct treatment is no longer possible.
- Sclerotia in substrates, pots and transportation boxes can survive for a long time and are easily spread in greenhouses in the water.
- Infested plants should be immediately removed from the stand, because of the high risk of infestation, and destroyed.
- In case of widespread Sclerotium infestation in greenhouses, the greenhouses must be thoroughly disinfected and hygiene measures have to be carried out.
Viroses
Biology
Viroses are very small and only visible under an electron microscope. They have no metabolism of their own
and only multiply in the cells of their hosts.
Viroses become more and more important in worldwide orchid cultivation, in Germany alone about 20 different viroses on orchids have been recorded. The symptoms of infestation depend on the family, species and
age of the infested plants and also on the conditions of cultivation. The viruses often occur in mixed infections
which makes a visual diagnosis even more difficult. Phalaenopsis hybrids are highly susceptible to these three
major viruses: the Cymbidium mosaic virus (CyMV), the Odontoglossum ring spot virus (ORV), tomato spotted
wilt virus (TSWV), impatiens necrotic spot virus (INSV) sowie das orchid fleck virus (OFV).
Cymbidium mosaic / Odontoglossum ring spot virus
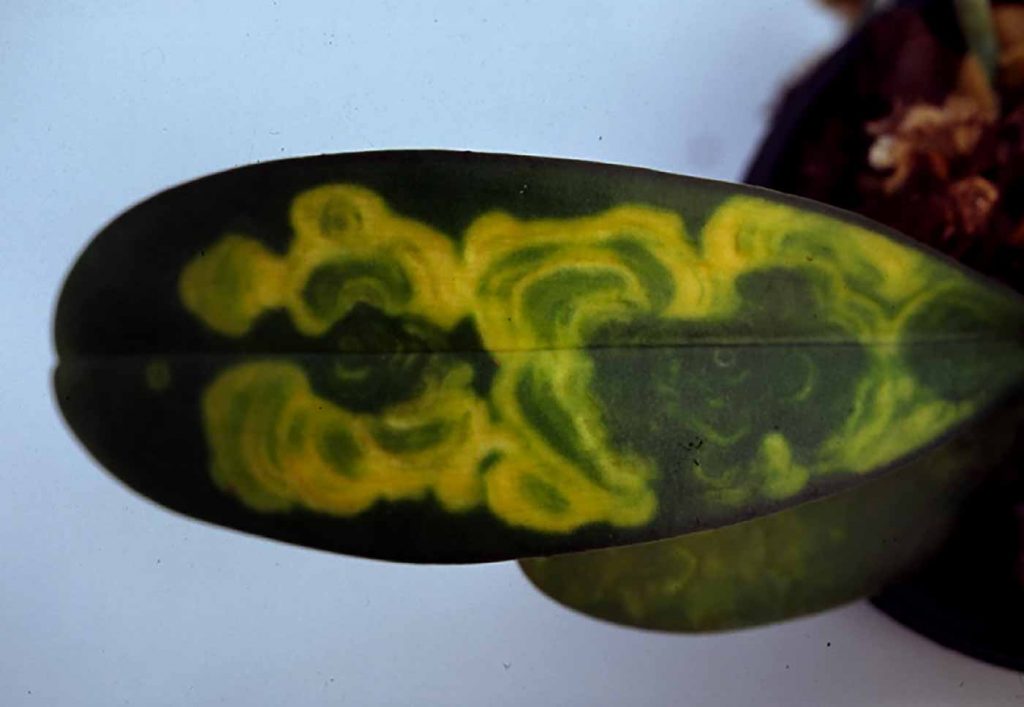
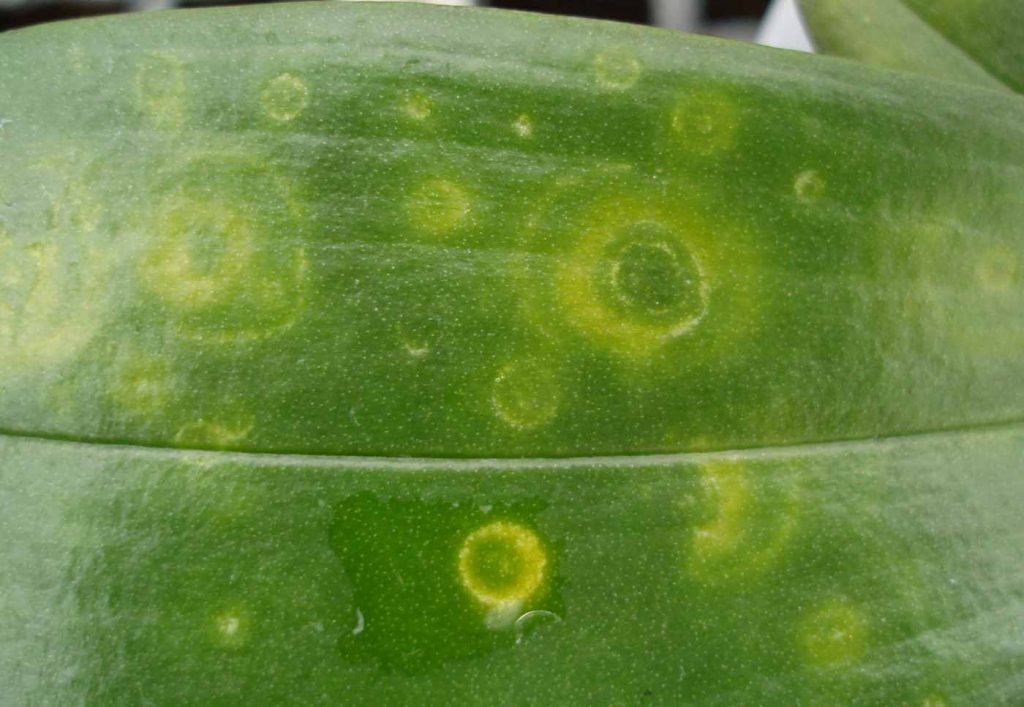
CyMV belongs to the potex virus group, ORV to the tobamovirus group. Viroses are clearly visible, especially
on younger leaves, by small or wide spots and often of a brighter tissue.
On Phalaenopsis, after ripening of the leaf, these bright areas change to sunken black spots, in the beginning
mainly on the undersides of the leaves, later also on the leaf surface.
In cases of severe infestation, the infested leaves drop off. On blossoms of dark-coloured varieties, marked
deformation and stripes or spotted areas are indicators of an infection with the Cymbidium mosaic virus, but
similar colour changes also happen due to mutations, mainly in dark Phalaenopsis varieties. Symptoms on blossoms are relatively rare on Phalaenopsis.
The young shoots of Cymbidium show small longish bright spots on the leaves, initially as longish stripes in the middle of the leaf, later on as marked spots on the leaf. With the disease progressing, older plants show black stripes on the upper and undersides of the leaf. Assimilation is considerably decreased, the leaves drop off. Blossom symptoms are frequent on Cymbidium.
Cattleya shows sunk in, dark brown to black discolouring on fully grown leaves. These discolourations may be found over the entire leaf surface or as round spots. New shoots are usually very bright. Small round brown spots can be seen on the open blossoms that join later to form brown stripes. Cattleya, Cymbidium and Phalaenopsis are the most susceptible orchid varieties to ORV and CyMV. However, these two viruses also occur on Odontoglossum and Laelia, but they are not frequently seen on Paphiopedilum.
Viruses can easily be transmitted mechanically, but there is no risk of sucking insects transmitting the virus. The risk of spreading the virus in water should not be underestimated. Both viruses are often found in mixed infections on orchids, attributing symptoms to one of the two viruses is hardly possible.
The young shoots of Cymbidium show small longish bright spots on the leaves, initially as longish stripes in the middle of the leaf, later on as marked spots on the leaf. With the disease progressing, older plants show black stripes on the upper and undersides of the leaf. Assimilation is considerably decreased, the leaves drop off. Blossom symptoms are frequent on Cymbidium.
Cattleya shows sunk in, dark brown to black discolouring on fully grown leaves. These discolourations may be found over the entire leaf surface or as round spots. New shoots are usually very bright. Small round brown spots can be seen on the open blossoms that join later to form brown stripes. Cattleya, Cymbidium and Phalaenopsis are the most susceptible orchid varieties to ORV and CyMV. However, these two viruses also occur on Odontoglossum and Laelia, but they are not frequently seen on Paphiopedilum.
Viruses can easily be transmitted mechanically, but there is no risk of sucking insects transmitting the virus. The risk of spreading the virus in water should not be underestimated. Both viruses are often found in mixed infections on orchids, attributing symptoms to one of the two viruses is hardly possible.
Tomato spotted wilt / Impatiens necrotic spot virus
Both viruses belong to the so-called Tospo viruses. In general, the leaves show slightly brighter spots, chloroses or cloudy spots. With the disease continuing, necroses and falling leaves appear and the plants die. Tospo
viruses are usually present locally in the leaf, but do not migrate systemically into the plant in orchids. The
major transmitter of Tospo viruses is the Western Flower Thrips (Frankliniella occidentalis Pergande).
In Asia, Capsicum chlorosis virus (CaCV) was also found on Phalaenopsis.
Orchid fleck virus
The orchid fleck virus usually appears on the older leaves of the plants of Phalaenopsis, Paphiopedilum, Cymbidium, Coelogyne, Oncidium and other orchids, whereas young leaves still look healthy. First symptoms on
the leaf surface are spots or stripes, with increasing infestation the tissue concerned becomes dry, there are
white-grey, clearly defined sunken spots, but not on the underside. Subsequently there is secondary fungus
infestation and the infested areas turn black. The virus can be latent in the plant without showing the abovedescribed symptoms. They may appear any time on the leaves, if the plant is exposed to stress. This virus is
transmitted by Brevipalpus californicus.
Control
Orchids are often latently infested, i.e. viruses are present in the plant, but without showing symptoms. When
they are transmitted to more sensitive species, symptoms may appear quickly. Stress or climatic and temperature changes may induce viruses from the latent to the symptoms phase.
- If a virus is suspected, examinations should be carried out in the laboratory.
- Viroses cannot be controlled chemically, thorough positive selection must be carried out in the greenhouse.
- Plants showing symptoms must be completely removed immediately, cutting off individual leaves is insufficient
- Sap transmission from plant to plant by touching, sprinkling water or work on the plants must be avoided.
- After each work process and most definitely after changing to a different species, hands must be washed thoroughly. Work on plants is the most common way of transmitting viroses in horticulture.
- For disinfection of cutting tools, tables, shelves and vessels, disinfectants can be used to destroy CyMV and ORV. The surfaces of cutting tools may also be cleaned with high percentage alcohol.
- The vectors of viruses, especially thrips and Individual viruses can also be transmitted by pests. The vectors, especially of thrips and aphids should be controlled consequently
Weeds & funghi in the substrate
Weeds in orchids
Ferns are among the most frequent weeds in orchids. They have similar needs to temperatures and humidity
as orchids and thus find optimal conditions where orchids are grown. Ferns can be distributed during pricking
or potting in greenhouses. Older plants form spores that are distributed in the spray water. To prevent that,
young ferns must be removed from the greenhouses.
Marchantia polymorpha L. grows mainly in nutrient-rich pot substrates, it is an indicator for high nutrient supply. Frequently, it comes together with the plants. Once established, the moss spreads when drops of water hit the so-called breeding cups. Marchantia polymorpha L. is very sensitive to higher pH values and dry substrate surfaces. Effective agents can be used that selectively effect the moss.
Creeping yellow wood-sorrel (Oxalis corniculata L.) is a soil-covering weed with green to dark red leaves and yellow flowers. The plant forms seed capsules from which the seeds are catapulted over a long distance. Therefore, the plant can spread quickly in the stand. As it also forms sprigs above and below the ground, it has good conditions on pot substrates. Creeping yellow wood-sorrel prefers pH values in the slightly sour range. Chemical treatment with herbicides is not possible in orchids.
Marchantia polymorpha L. grows mainly in nutrient-rich pot substrates, it is an indicator for high nutrient supply. Frequently, it comes together with the plants. Once established, the moss spreads when drops of water hit the so-called breeding cups. Marchantia polymorpha L. is very sensitive to higher pH values and dry substrate surfaces. Effective agents can be used that selectively effect the moss.
Creeping yellow wood-sorrel (Oxalis corniculata L.) is a soil-covering weed with green to dark red leaves and yellow flowers. The plant forms seed capsules from which the seeds are catapulted over a long distance. Therefore, the plant can spread quickly in the stand. As it also forms sprigs above and below the ground, it has good conditions on pot substrates. Creeping yellow wood-sorrel prefers pH values in the slightly sour range. Chemical treatment with herbicides is not possible in orchids.
Funghi in the substrate
Leucocoprinus birnbaumii ([Corda] Sing.) is a saprobiontic soil inhabitant. The fungus is found all over the
world in tropical areas where it lives on dead, decomposing organic substances. As the fungus loves humidity
and heat, it is often brought into greenhouses where it finds ideal conditions in pot substrates.
Noticeable is the citrus-yellow, 3-5 cm long fruit body. At first it looks like a small cone, later on it opens, dries
and gets a greyish colour. The major problem are the whitish sclerotia of the fungus that are ca. 1mm in diameter. In particular, they penetrate through the transparent pots of Phalaenopsis. This considerably reduces the
decorative value of the orchids. Due to its water-repellent properties, wetting of the substrate is also made
more difficult.
Only hygienic measures help against spreading this fungus. All pots with fungus phases must be disposed of. Direct treatment of the fungus is hardly possible. Early use of fungicides against the spores is the only way to improve the situation.
Only hygienic measures help against spreading this fungus. All pots with fungus phases must be disposed of. Direct treatment of the fungus is hardly possible. Early use of fungicides against the spores is the only way to improve the situation.